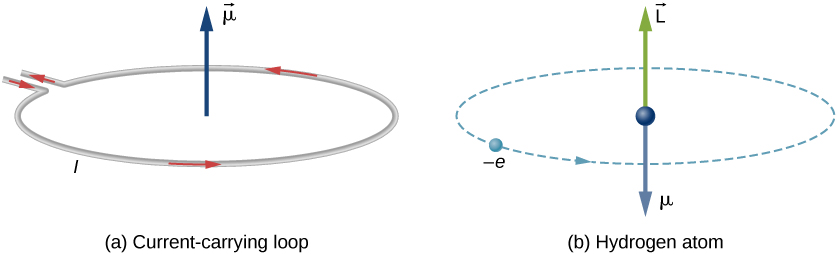
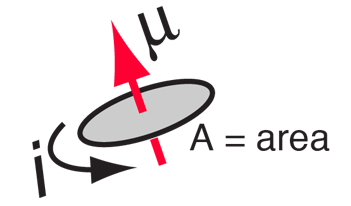
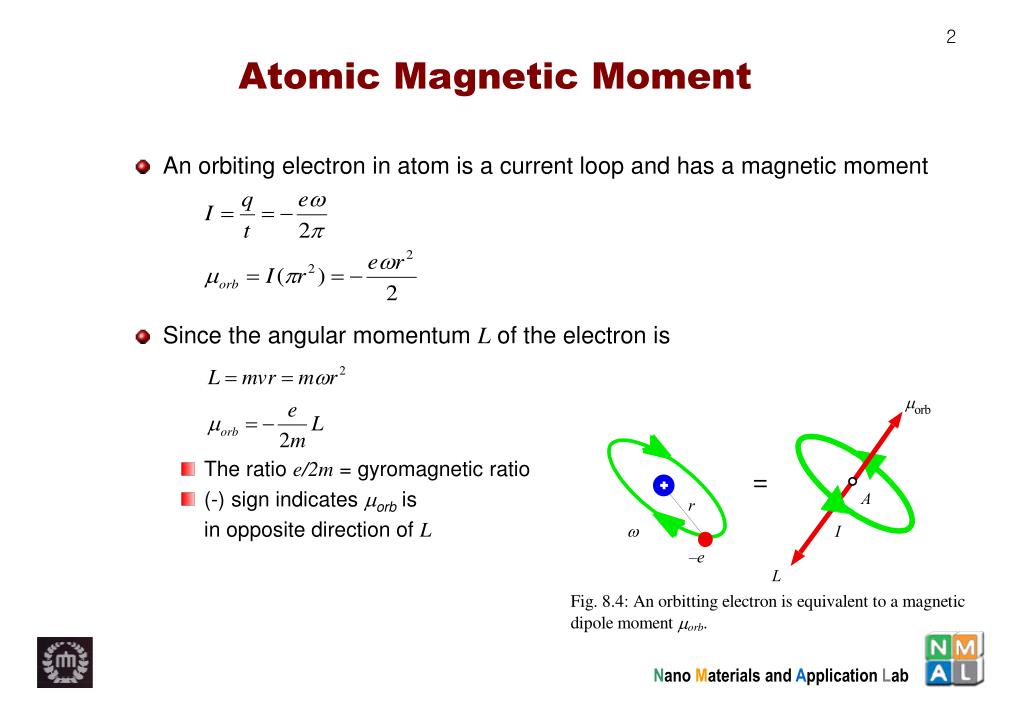
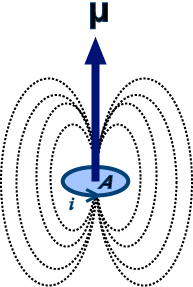
SOLVED:The magnetic dipole moment of the iron atom is about 2.1 \times 10^{-23} \mathrm{A} \cdot \mathrm{m}^{2} . (a) Calculate the maximum magnetic dipole moment of a domain consisting of 10^{19} iron atoms. (

Calculate the change in magnetic dipole moment when the electron in a H-atom makes a transition from the n=4 orbit to the n=3 orbit. Suppose that the atoms is in a magnetic

Three protons coming from excited atomic hydrogen sample are picked up. Their energies are 12.1 eV, 10.2 eV and 1.9 eV . These photons must come from

If the magnetic dipole moment of an atom of diamagnetic material, paramagnetic material, and ferromagnetic material are denoted by mu d, mu p and mu f respectively, then




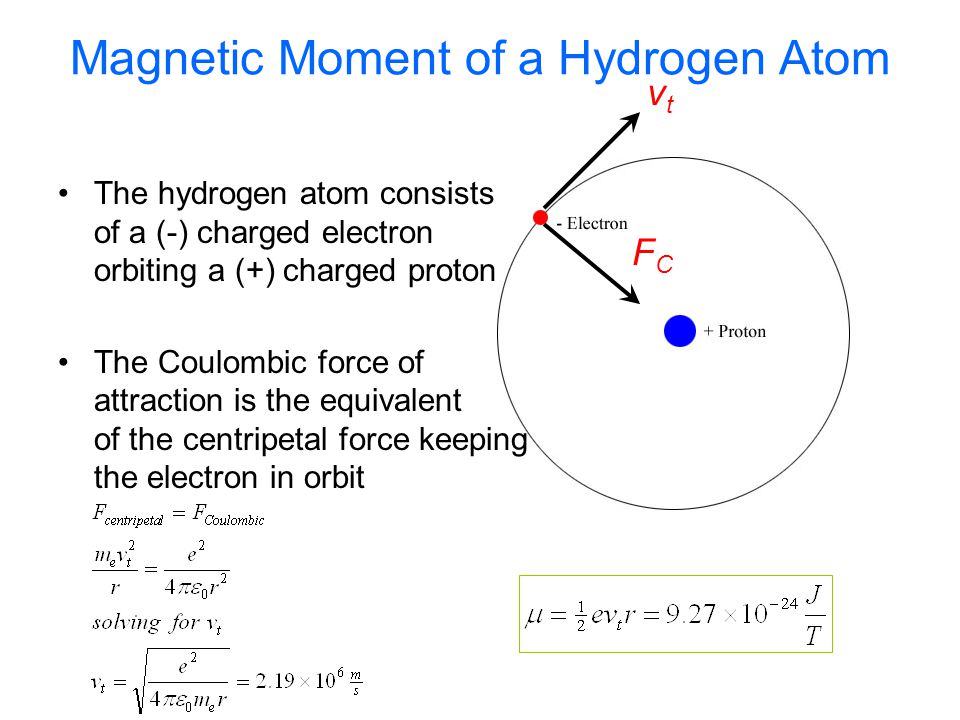


.PNG)