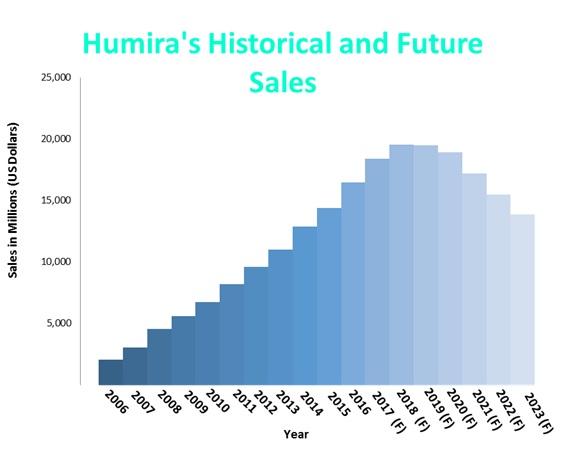
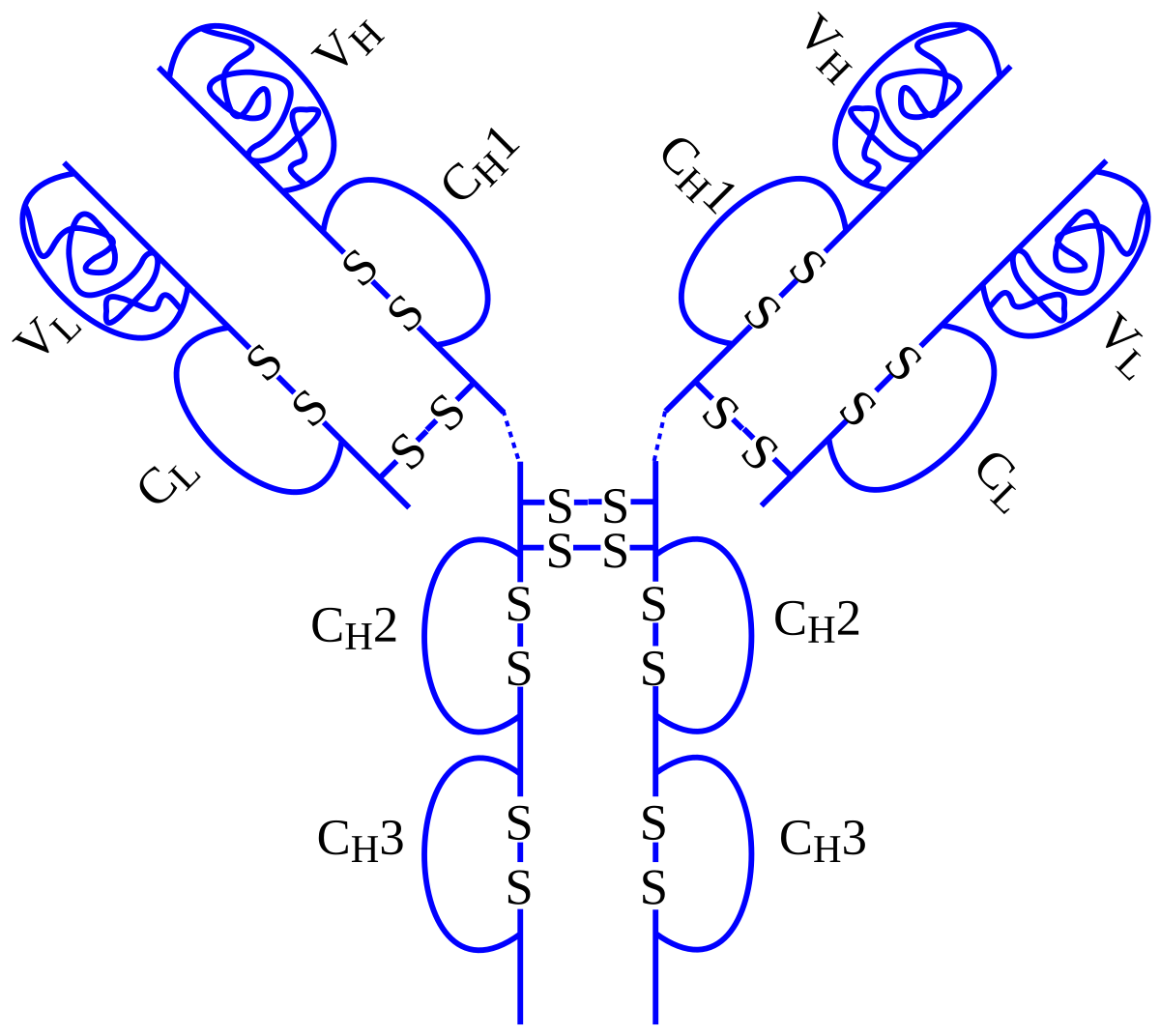
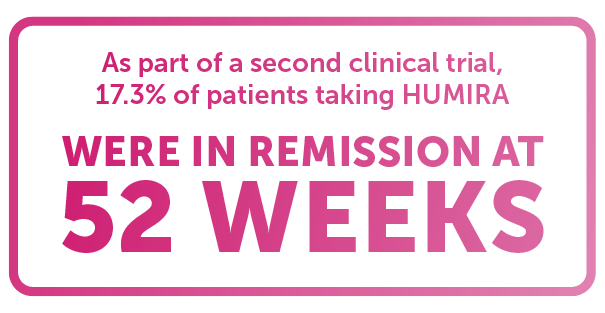
تويتر \ Dr T ² | 🙈 🙉 🙊 على تويتر: "Found this gem while going through old records: denial of Humira for #Crohns because it was deemed experimental. Forced me into

Adalimumab in Japanese patients with active ulcers of pyoderma gangrenosum: Twenty‐six‐week phase 3 open‐label study - Yamasaki - 2020 - The Journal of Dermatology - Wiley Online Library

Long-Term Safety of Adalimumab (HUMIRA) in Adult Patients from Global Clinical Trials across Multiple Indications: An Updated Analysis in 29,987 Patients Representing 56,951 Patient-Years - ACR Meeting Abstracts
![PDF] Comparison of the Efficacy and Safety of Adalimumab (Humira) and the Adalimumab Biosimilar Candidate (HS016) in Chinese Patients with Active Ankylosing Spondylitis: A Multicenter, Randomized, Double-Blind, Parallel, Phase III Clinical Trial PDF] Comparison of the Efficacy and Safety of Adalimumab (Humira) and the Adalimumab Biosimilar Candidate (HS016) in Chinese Patients with Active Ankylosing Spondylitis: A Multicenter, Randomized, Double-Blind, Parallel, Phase III Clinical Trial](https://d3i71xaburhd42.cloudfront.net/1f3ea24e842b7750dfbe6b098b7b1b52e074980f/5-Figure1-1.png)
PDF] Comparison of the Efficacy and Safety of Adalimumab (Humira) and the Adalimumab Biosimilar Candidate (HS016) in Chinese Patients with Active Ankylosing Spondylitis: A Multicenter, Randomized, Double-Blind, Parallel, Phase III Clinical Trial

Adalimumab for Maintenance of Clinical Response and Remission in Patients With Crohn's Disease: The CHARM Trial - Gastroenterology

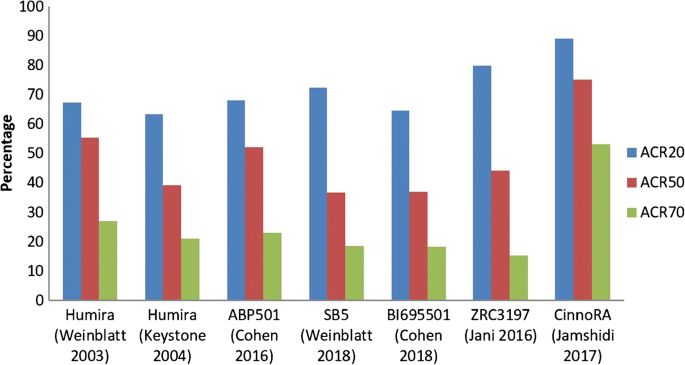
Phase III clinical trials conducted for adalimumab biosimilars; data... | Download Scientific Diagram

Tocilizumab monotherapy versus adalimumab monotherapy for treatment of rheumatoid arthritis (ADACTA): a randomised, double-blind, controlled phase 4 trial - The Lancet

Risankizumab compared with adalimumab in patients with moderate-to-severe plaque psoriasis (IMMvent): a randomised, double-blind, active-comparator- controlled phase 3 trial - The Lancet
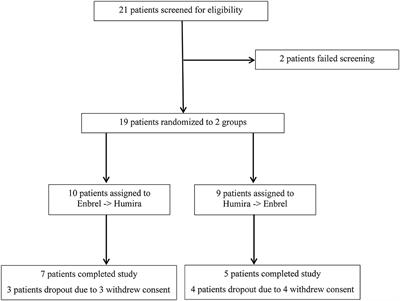
Frontiers | Head-to-Head Comparison of Etanercept vs. Adalimumab in the Treatment of Ankylosing Spondylitis: An Open-Label Randomized Controlled Crossover Clinical Trial | Medicine