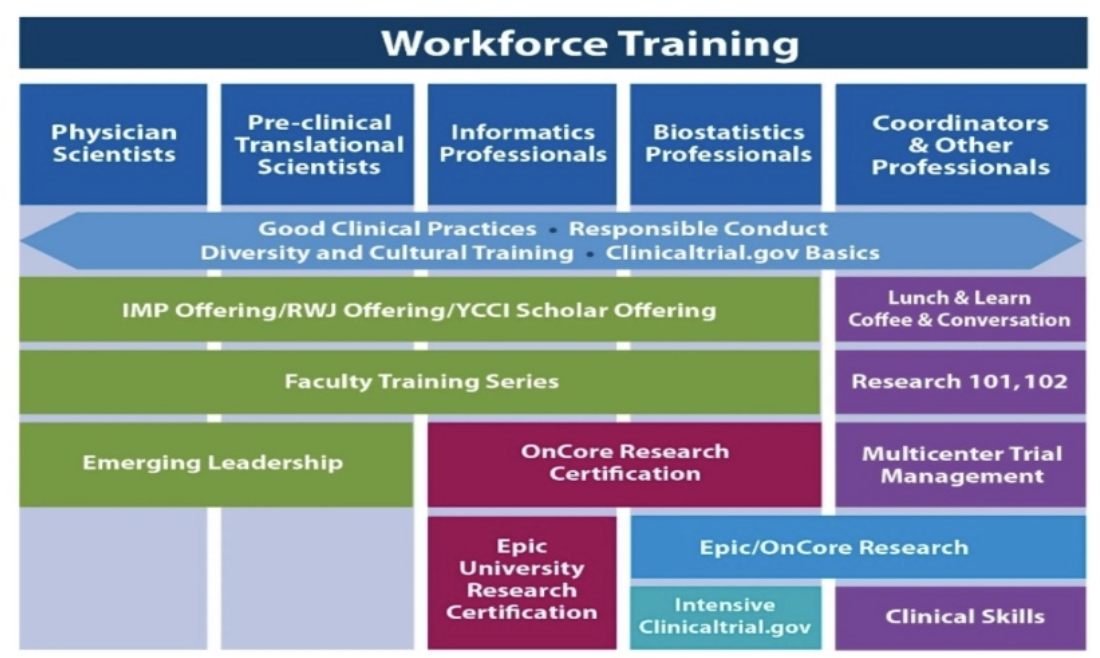
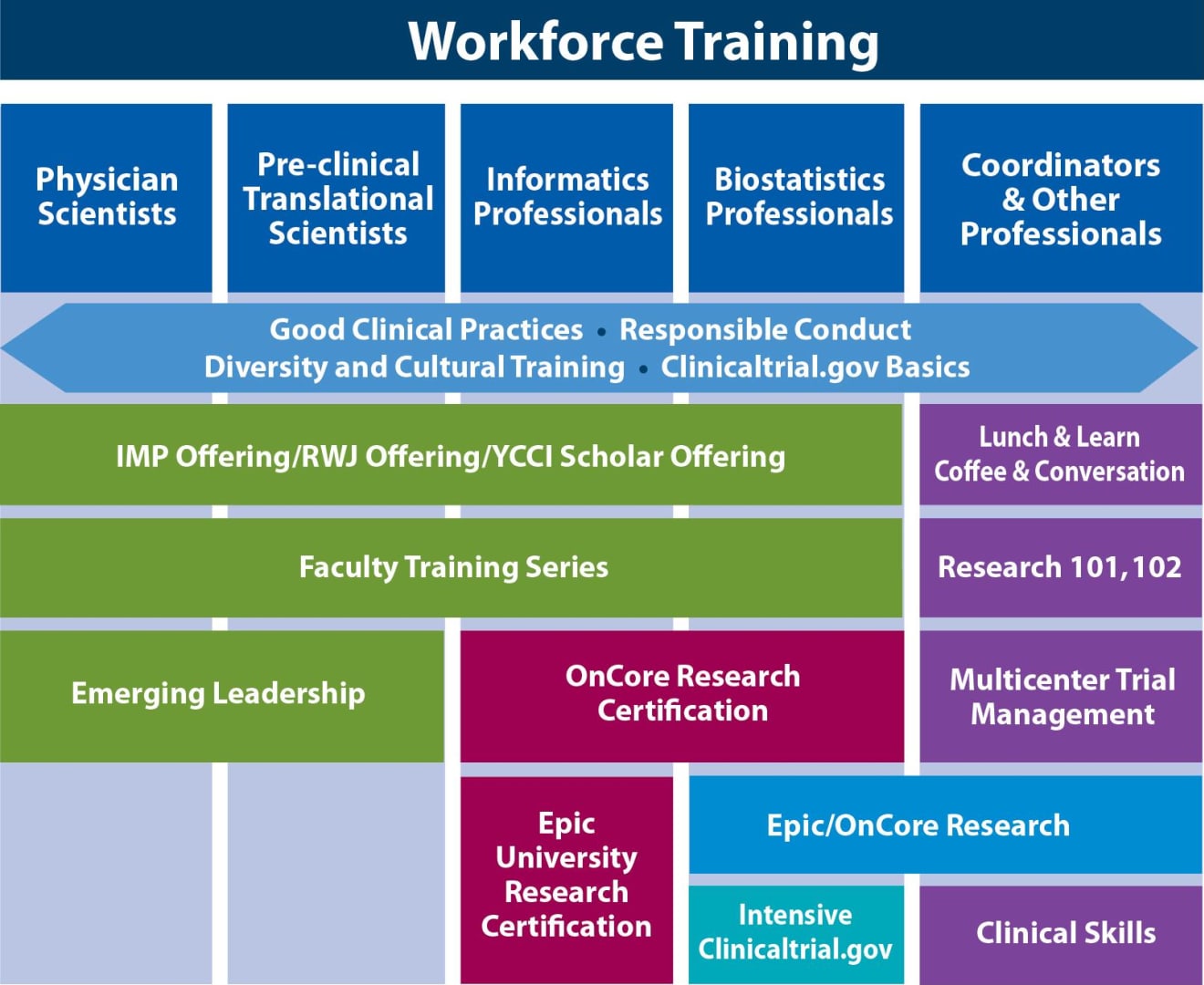
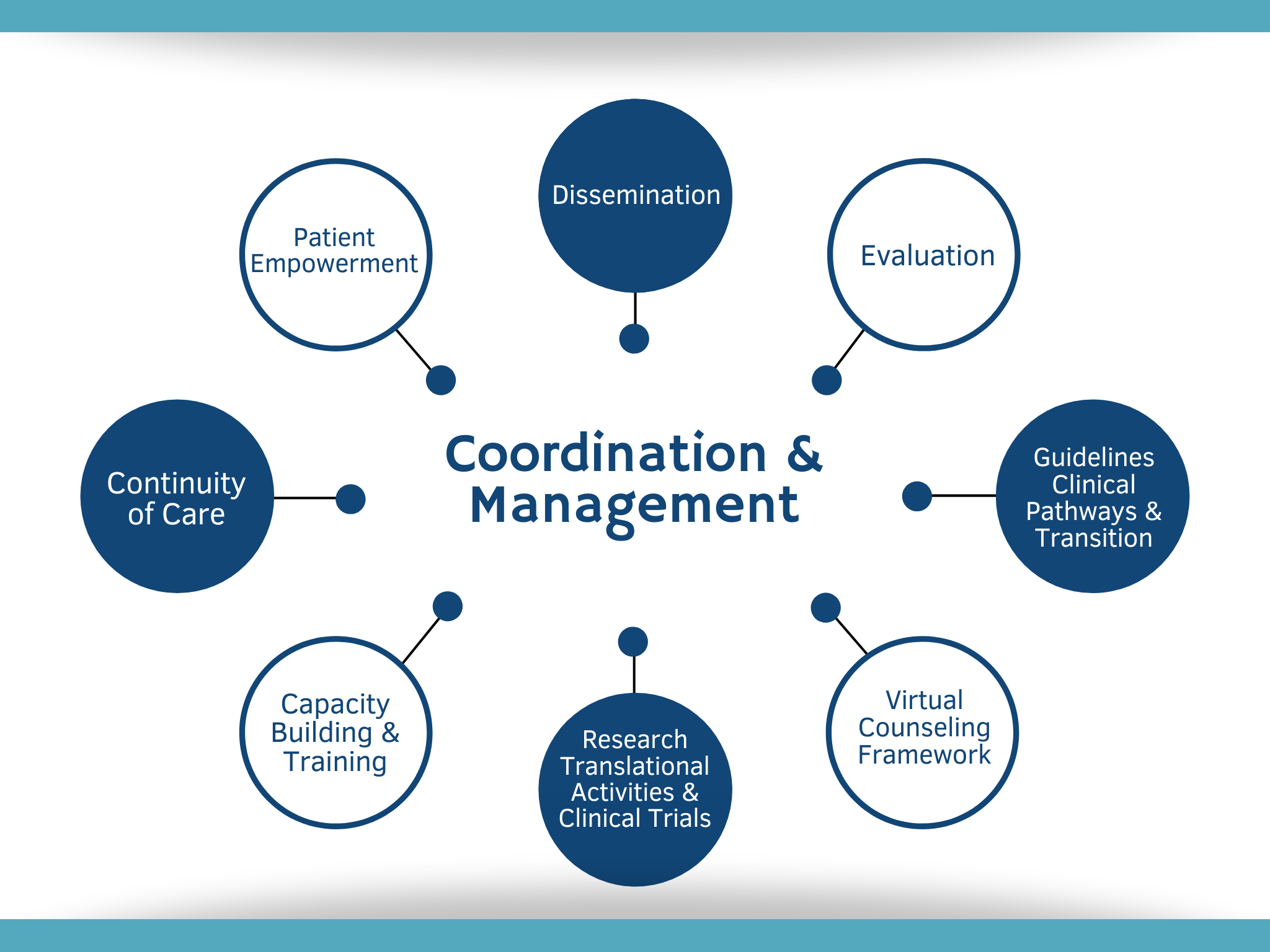
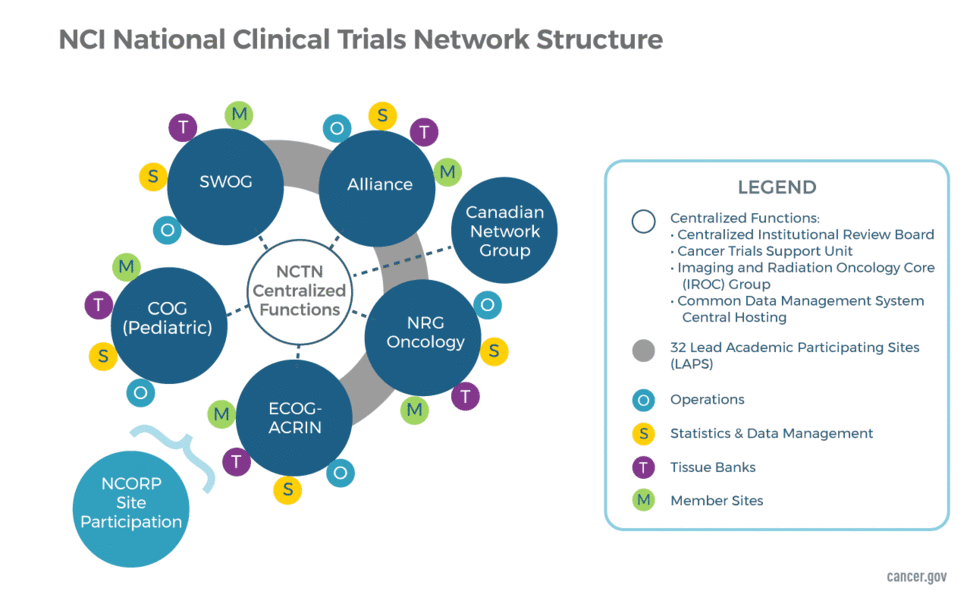
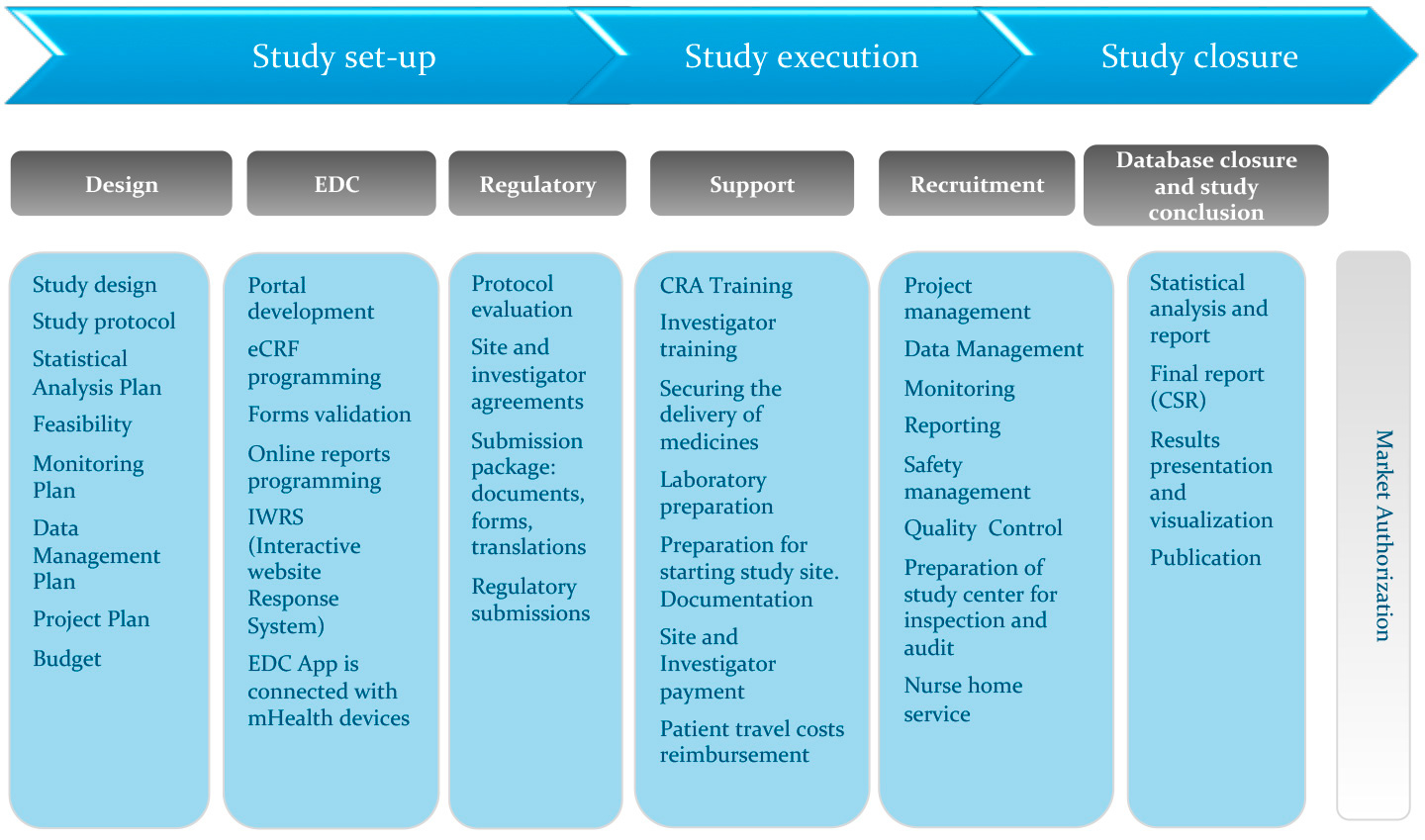
Clinical Trials Management Certificate — online course - University of Delaware Div. of Professional and Continuing Studies

Good Clinical Practice (GCP) training: Helping you find your next clinical research job | Proclinical Blogs

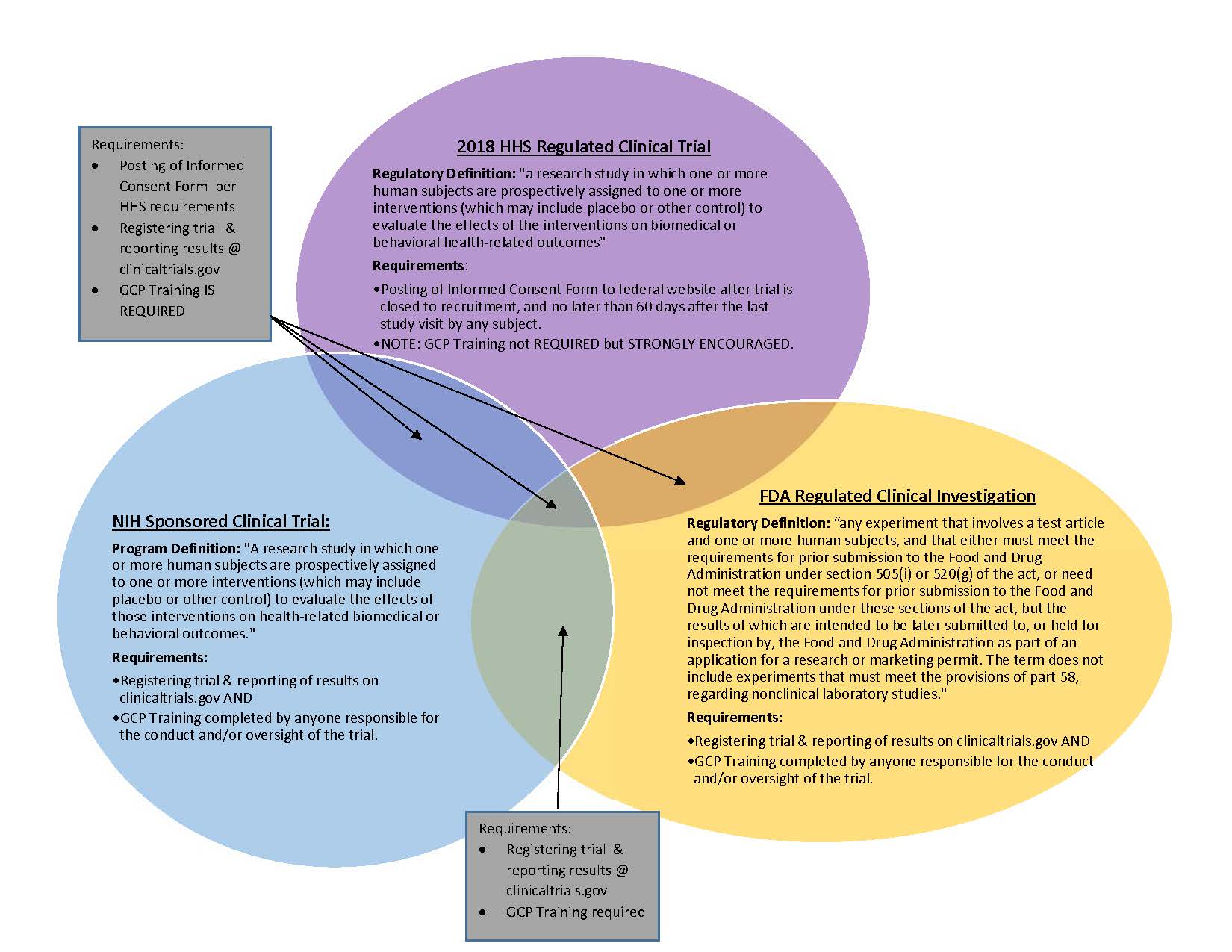
Atlanta Pediatric Research | NIH Requirements for Human Subject Research | Clinical Research Resources | Research Resources | Research | Emory + Children's + GT | Atlanta Pediatric Research Alliance
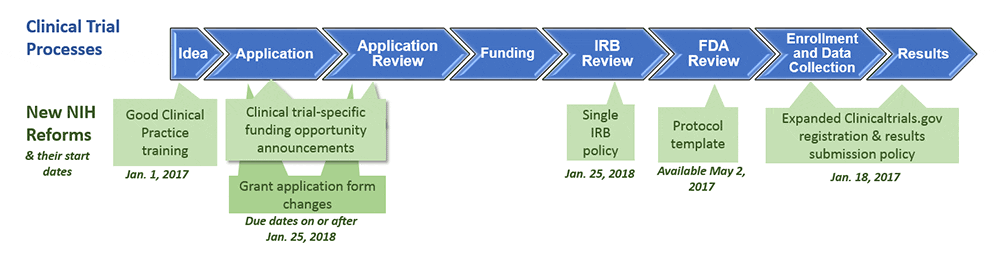
NIH Redefines Clinical Trials and Sets New Requirements: Is Your Human Subjects Research Affected? - ASHG